SWPRS | 18 Dec 2021
New insights into coronavirus disease and treatment.
Please note: Patients are asked to consult a doctor.
What is severe covid, really?
In most people, covid is a rather mild or even asymptomatic infection: according to recent studies, about one third of people who test positive never show any symptoms. The overall hospitalization rate is about 1% to 3% and very strongly depends on age and prior health status: it is much lower than 1% in children, but higher than 10% in the elderly.
Thus, the real question is: what is severe covid, and how can it be prevented?
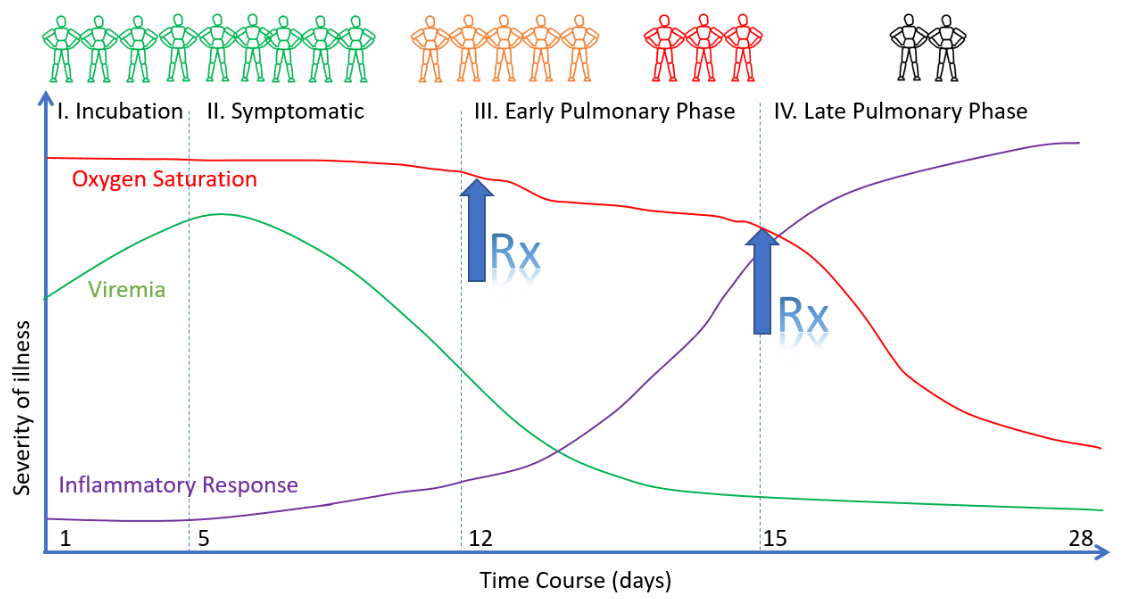
A fascinating new preprint study by researchers at the University of Pennsylvania found that, in mice, SARS-CoV-2 did not have to infect the lungs at all to cause acute lung injury (ARDS), hypoxemia (low blood oxygen), and death. Instead, an infection of the olfactory bulb in the nasal cavity was sufficient to cause hypoxemia and fatal covid lung injury.
This very intriguing result is consistent with earlier findings, previously discussed by SPR, that severe covid appears to be not just a viral pneumonia, but a post-viral auto-immune attack on lung tissue, causing hypercoagulation, thrombosis, and respiratory failure. Specifically, severe covid might be an anti-phospholipid antibody syndrome (APS) targeting annexin A2.
This might explain why some covid patients experience a sudden and rapid deterioration about 7 to 10 days after initial symptom onset, when viral load has already decreased substantially in most people. A post-viral, hyper-inflammatory auto-immune attack may also explain why treatment with corticosteroids is successful against severe covid, whereas monoclonal antibodies fail. It may also explain why, in many cases, simply putting covid patients on a ventilator was such a bad idea.
With covid vaccines providing only short-term protection and severe covid being very difficult to treat, effective early treatment options for high-risk patients should remain a top priority.
Early treatment: an update
Ivermectin
All of the randomized controlled trials showing a significant mortality benefit of ivermectin turned out to be fraudulent. The epidemiological evidence doesn’t support a strong effect of ivermectin, either: in the Indian state of Uttar Pradesh, for instance, the total death count was about 43 times higher than the official covid death toll.
However, two high-quality RCTs of ivermectin are still ongoing, and ivermectin recently passed the “futility threshold” in both of these trials. Thus, it is still possible that ivermectin may turn out to offer some moderate immuno-modulatory benefit against covid.
Molnupiravir (the Merck pill)
Merck initially claimed that its covid pill, Molnupiravir, is highly effective against covid. But the full study data shows that only the Brazilian arm of the trial showed a significant benefit, and against the Delta variant, Molnupiravir performed worse than a placebo.
In addition, Molnupiravir’s mode of action may be toxic to human DNA and may drive dangerous coronavirus mutations. In sum, Molnupiravir doesn’t look like a good choice, and French authorities have already rejected authorization of the drug.
Paxlovid (the Pfizer pill)
Pfizer has not yet released their study data, but wrote in a press release that Paxlovid reduced hospitalizations and deaths in at-risk groups by nearly 90%. Paxlovid directly targets the main coronavirus replication enzyme (3CLpro), which certainly is a promising approach.
However, Paxlovid will only be widely available by mid-2022, and it will be quite expensive. One may also ask if people receiving such a powerful anti-viral drug within days of symptom onset will develop full immunity against the virus, or if they will remain prone to reinfection.
It has been noted that Paxlovid and Ivermectin both inhibit the same coronavirus replication enzyme (3CLpro). This is correct, but Paxlovid is about 1000 times more effective, whereas Ivermectin requires a dosage that is unlikely to be achievable in humans.
Monoclonal antibodies
Monoclonal antibodies have been shown to be effective in early treatment but ineffective in the treatment of already hospitalized patients (post-viral phase). In addition, monoclonal antibodies have rapidly lost efficacy against new viral variants, and especially against omicron.
As an example, Germany had bought 190,000 doses of monoclonal antibodies at a cost of 400 million euros, but used only 8,000 doses, donated 70,000 doses to other countries, and recently had to dispose of the remaining doses. A colossal public health failure and waste of public money.
Budesonide (an inhalable asthma drug)
In early 2021, two British studies found that early treatment with budesonide (an inhalable steroid asthma drug) was effective at preventing disease progression and hospitalization. SPR at the time noted that budesonide improved soft endpoints but failed to improve hard endpoints, consistent with a drug that has only a superficial effect on symptoms, not on the disease.
A recent trial of a similar inhalable steroid drug, published in JAMA, came to a similar mixed result, but unfortunately included mostly young and low-risk participants.
In sum, budesonide may help alleviate symptoms of mild to moderate covid, but it remains uncertain if it actually can prevent disease progression and severe covid.
Fluvoxamine
The TOGETHER trial found that early treatment of covid patients with fluvoxamine (an anti-depressant impacting serotonin metabolism) reduced covid “hospitalizations or ER visits” significantly by 32%. But the trial has been criticized as fluvoxamine failed to reach hard endpoints (like hospitalization or mortality) and endpoints had been changed during the trial.
It is thought that fluvoxamine may work against covid as an anti-inflammatory drug or by targeting platelet serotonin metabolism. In the latter case, cyproheptadine, a direct serotonin receptor antagonist, may be even more effective.
Anti-androgen treatment
SARS-CoV-2 uses the ACE2 cell receptor, the expression of which is driven by androgens (male sexual hormones). Early studies found that older men receiving anti-androgens – typically used against prostate cancer or hair loss – were at a much lower risk of severe covid.
Studies by a Brazilian group found that a specific anti-androgen drug was highly effective against covid, but these results have been strongly criticized by other researchers and they have not yet been replicated. Results of a US phase II/III trial should be available soon.
Artemisia annua (tea, powder, or extract)
Pre-clinical and early clinical studies, including an in vitro study by the German Max Planck Insitute, found that artemisia annua, a plant used against malaria, may be effective against covid.
Intriguingly, the one country that has relied on artemsia annua in its fight against covid – Madagascar – continues to have one of the lowest excess mortality rates in the world.
Arginine (an amino acid)
Arginine is an amino acid vital to the immune system, endothelial function and tissue repair. Studies found that arginine depletion is a major predictor of severe and fatal covid. A small Italian RCT found that arginine significantly improved the recovery of covid patients.
For a recent review, see l-Arginine and COVID-19: An Update (Nurtrients, November 2021).
Aspirin and heparin
Available studies on early or prophylactic treatment with aspirin or heparin are mostly positive, whereas results in hospitalized patients are mixed or negative. Aspirin and heparin act as antiplatelet and anticoagulation drugs (to prevent platelet hyper-reactivity and covid thrombosis).
Vitamin D and vitamin C
Vitamin D plays an important role in the immune response to infections. Avoiding vitamin D deficiency, especially in the elderly, is essential. Once infected, it is too late to take standard vitamin D (which requires about ten days to be metabolized into its active form). However, in patients with vitamin D deficiency, administering a high dose of biologically active vitamin D (calcifediol or even calcitriol) may be effective.
A recent review of trials on vitamin C against covid found that in-hospital intravenous vitamin C intervention “may improve oxygenation parameters, reduce inflammatory markers, decrease days in hospital and reduce mortality”. In contrast, the evidence on the use of oral vitamin C supplements was insufficient.
Hydroxychloroquine (HCQ)
HCQ, known as an anti-malaria drug, is also a standard drug against some chronic auto-immune conditions, including anti-phospholipid antibody syndrome (APS). Thus, if severe covid turns out to be a form of acute APS, HCQ might, after all, be helpful as an early or prophylactic treatment to prevent severe covid in high-risk patients.
Previous negative trials of HCQ mostly investigated anti-viral properties or they were run in hospitalized patients or in young and low-risk patients. Such trials were bound to fail. In contrast, a prospective cohort study in Saudi Arabia found that HCQ was associated with a 43% reduction in hospitalizations and 54% reduction in deaths. A very large retrospective study in Iran found that HCQ was associated with a 38% reduction in hospitalizations and a 73% reduction in deaths.


